We specialize in catheter designs and process development (manufacturing), low volume prototype production, additive manufacturing/3D prototype development, laser weld (spot welds) and laser cutting processes, balloon catheter mold forming, test systems automation using LabView & C+ programs and equipment validation – GAMP5 (Ovens – Thermal Mapping, Drop Tester, Vibration Tester, Mechanical Testers). We are ISO-13485 & 21 CFR Part 11 Compliant. 95660 | Phone: 916.993.3799 | Email: Inc., a privately owned consulting company with over 25years of experience serving the biotech and medical device industry. PIL&M Inc., 3437 Myrtle Avenue #355- north highlands, ca. 
Revalidate as required if there are any changes to the computer systems or softwareįor further assistance regarding how to validate your new or existing equipment computer software/systems or commissioning contact us.Validated computer systems must be retained in a revision-controlled data archive application.Release the computer system validation report including all pertinent documents that may have been created. 
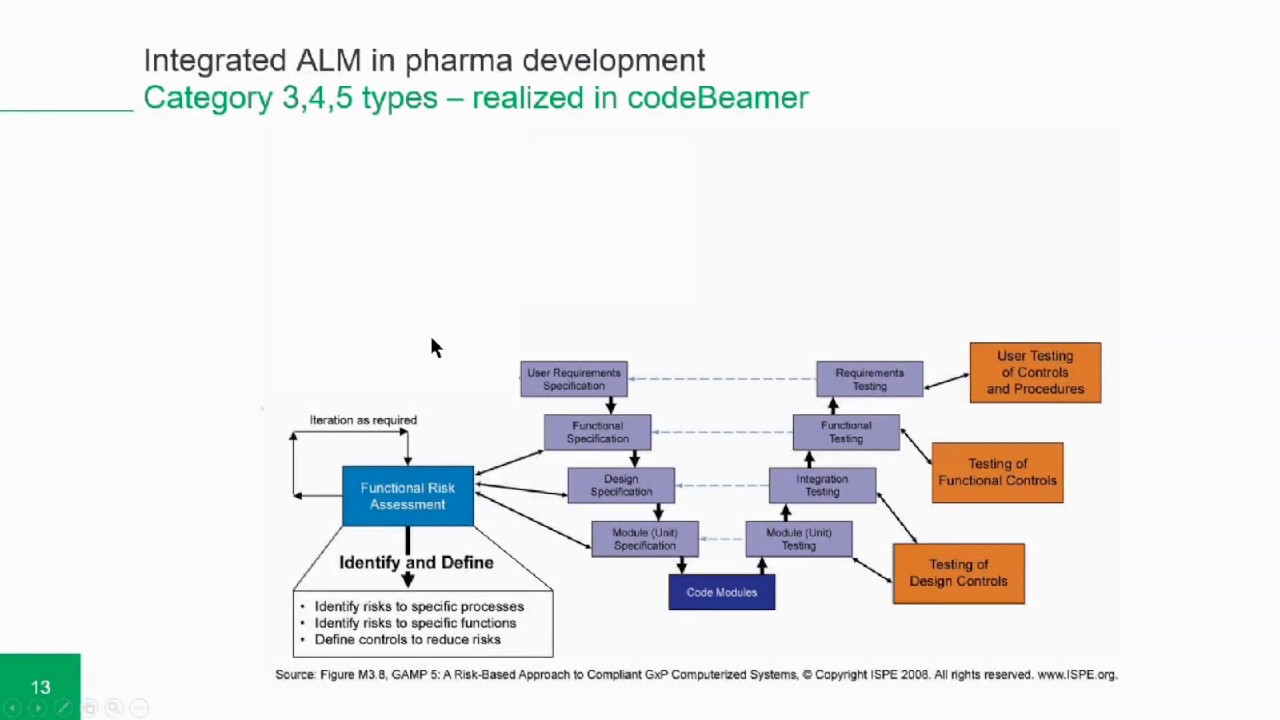
Update the software configuration section of the computer system validation report.Document any deviations taken from the plan in the deviation section of the computer system validation report.Document all the test cases result in the computer system validation report.Execute all documented test cases per the computer system validation plan/protocol.Identify the safety, compliance, environmental impact, ergonomics and business risks.Capture all pertinent validation deliverables on a computer system validation form.Based on the selected computer system category, validation must be performed per the following deliverables – FAT, SAT, FS, URS, IQ, OQ, PQ, Risk Hazard Analysis.Note: if the answer to the above questions are NO, the computer software system does not have to be validated. Category 5: Custom computer software system developed to meet individual user requirements, where there is no commercial Computer software System available.Category 4: Computer software that are configured for a specific business operation.Category 2/3: Products which are used Off-The-Shelf (OTS) or without software.Category 1: Infrastructure Computer Software.Maintenance of calibration records or tracking of calibration conditions.To demonstrate compliance to a regulation.Determine if computer system is new or previously validated.The purpose of the computer system e.g.:.Used during a development of a new product or manufacturing.Identify the equipment used in the manufacturing process.Through means of proper documentation and traceability each equipment is validated as follows: Computer System Validation (CSV) – GAMP5 (Good Automated Manufacturing Practice V-Model), is a guidance per FDA 21 CFR Part 11-820 & IS0-13485.75, that all Biotech and medical device companies usually follow to ensure that their respective testing or manufacturing equipment functions and performs as intended or expected. Any computer-based equipment or fixture that is required for testing or the production of clinical or human use devices must be validated prior to use.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
